|
Many test methods are available for testing various customers' products with different bacterial and virus strains. This EN 1276 is one of the bacterial testing methods that help to find the bacterial activity on various chemical products like antiseptics and disinfectants. This test is necessary because these chemical components are for medical use, so the users should be confident. So, labs conduct these tests on the products to check the ability of the products against the bacteria. And there are many other tests also available for different products.
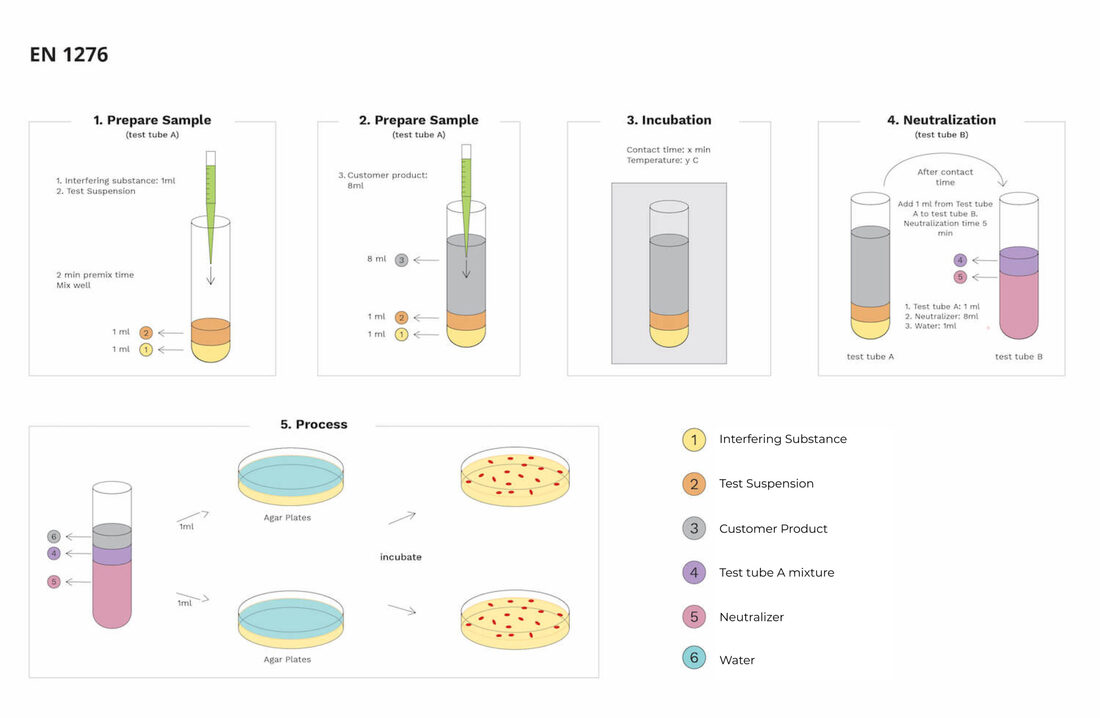
Antibacterial testing methods Laboratories have various antibacterial testing methods for plastic, textile materials, and even cleaning agents like detergents. Each antibacterial testing method uses real-life bacteria, which helps to develop the bacterias on the sample products. The bacterias available in this test method are some bacterial organisms like Staphylococcus aureus, Pseudomonas Aeruginosa, Enterococcus, and Escherichia coli. Several antibacterial testing methods like ASTME 2149, JIS L 1902, JIS Z 2801, AATCC- 100 are available for various products. These test methods are for different products, and each test method will have a unique testing process to find the output. Similarly, many antiviral testing methods are available for various products, which works equally to antibacterial testing. Testing process The test process of this EN 1276 contains various steps, and each step of the test has a unique rule for processing the test correctly. While performing these antibacterial and antiviral tests, the rules are essential because a small mistake in regulations may change the entire test result. It can also lead to the destruction of samples and strains. This test consists of 4 steps Sample preparation Incubation Neutralization Process Sample preparation The test starts in this process with a clean test tube, and in that test tube, they add 1ml of interfering substance and 1ml of test suspension. It is the first set of sample preparation, and in the second set, they pour 8 ml of customer product into the existing solutions. All these are the sample preparation process, and the name of this test tube is A. Incubation & Neutralization Test tube A will be under incubation for a specific time at a particular temperature in the incubation process. The time and temperature will differ as per the product. In the Neutralization process, test tube B will get 1 ml of test tube A solution and 8 ml of neutralizer along with 1 ml of water. Final Process In this process, the final test tube B is under testing and from that test tube, they take 1ml for each agar plate. Then the process will continue in 2 agar plates. Then the agar plates are under incubation for creating the final output. These are the test method of EN 1276. After completing all the steps in the test method, the final result will explain the detail about the ability of the antiseptics and disinfectants. Conclusion These are the testing method of EN 1276, and this test method is the best option for testing chemical materials like antiseptics and disinfectants. All these antibacterial and antiviral testing methods will help to find the abilities of all the customer products.
0 Comments
The EN 1276 test specification defines a suspended test to determine whether a chemical antimicrobial agent or disinfectant is bacteriostatic. Antimicrobials or antibacterial products are meant to control and combat specific populations of harmful microbes.
This antimicrobial method considers reasonable circumstances and issues of interest substances in modeling the actual implementation of the product. Except in cases where disinfection is medically appropriate, the test was applied to formulas or bioactive molecules used in food, manufacturing, household, as well as organizational environments. Whereas the method is not typically applicable to beauty products, it could be used in the noted areas of utilization for those intended for hand washing. What Exactly Is Disinfection? Disinfection refers to the process of removing many or even all pathogenic microorganisms from various objects, excluding microbial cells. Items in health-care settings are typically disinfected using chemical products or wet pasteurization. Products That Are Testable? When mixed with distilled water or, inside the case in order to maximize the performance, chemical cleaning agent and antiseptic intended to represent a relatively homogenous, thermodynamically stable preparation. It means that goods in use in snacks, industrial, domestic, and organizational settings, excluding regions and circumstances where disinfection is physician indicated, by excluding merchandise in use on having lived tissues other than those used for hand hygiene in the above-mentioned settings. This EN 1276 test means that goods in use in snacks, industrial, household, and organizational settings, excluding regions and circumstances in which disinfection is physician indicated while excluding product lines in use on living tissues other than those used for hand hygiene in the above-mentioned settings. At the very least, the following aspects are cover: a) manufacturing, distribution, and retailing of: Animal-derived foods: milk and milk products, beef, fish, sushi, and similar items, eggs and dairy goods feed for animals, and so on. Food of plant origin: liquors; fruits, veggies, and derivative products (including sugar, single malt, and so on) flour, puttering, and making bread; animal feeds and so on. b) organizational as well as domestic areas: designed to cater institutions; common areas; public transit; schools; nursery schools; stores; sporting events rooms; end up wasting bins (bins, etc.); guesthouses; dwellings; medically shared by users areas of hospitals; office buildings; and so on. c) Other manufacturing areas: packaging materials; biotechnology (yeast, peptides, enzymes, and so on); pharmaceutical; personal care products; textiles; space program, games industry, and so on. In the food service and farming processing, in which hygiene is critical and clear rules are in place, the cleaning solutions you utilize must meet all of the required standards. Picking the correct product necessitates careful thought, not just for the job at hand, as well as for the tier of decontamination needed in your surroundings. Preparing food floors as well as equipment that comes in contact with food can become polluted with a variety of bacteria. The en 14476 test is a test method that helps to detect virus activities that are taking place on a particular material. This is a method that uses the help of the standard European method that also helps to detect the coronavirus in the tested area. Well, this method is also denoted as the key method to detect coronavirus. There are a number of benefits for which most of the pathologies are taking help from this method, and European countries have also detected it as the standard method of testing. It is an efficient method that will also allow you to get effective results in any situation.
1. It will follow EU guidelines. It will remain helpful for the testers as this one will follow the EU guidelines. As a result, it will generate the quickest results for the experts and the experts will check the accuracy of the results whenever taking help from this test so that the test methodology can be taken place with the help of the test. 2. It is more reproducible. When you are taking help from the en 14476 test, you will be able to make sure that the results are reproducible. As a result, this one can say that this one is the most accurate factor that would be effective for each and every stage. Especially on the hard surface, it will give the most effective results in the detection of viruses. The results will generate the most authentic and accurate results on the hard surface to get the most reproducible results on the surface. As a result, it also helps in generating the quickest results that are the primary reasons Europe has decided it as the standard method to test. 3. Ingredient concentration detection While taking help from the en 14476 test, you will be able to detect the concentrations that are being used in the test. This is another thing that the tests can generate, and for this reason, most of the pathologies also take help from this test. 4. Helps in product dilution This methodology acts for product dilution caused by the use of interfering substances and viral nutrient broth, avoiding artificially reduced product efficacy. This is probably another reason why most pathologies are taking help from the product delusion. Conclusion Here are the reasons for which the importance of the en 14476 test is repeatedly increasing. These tests will also help the pathology detect the appearance of the microbe in the field of the surface. As a result, this one has also become the most effective step by which the testing would complete in any situation. Also read - EN 1276 – for quantitative testing on disinfectants and other similar products It is necessary to use viable isolation of HSV in order to conduct the susceptibility assay. Antiviral Testing Lab Clinicians should obtain clinical specimens from the cutaneous, vaginal, oral-facial, or other suitable body areas if necessary and send them to a diagnostic laboratory for viral isolation if necessary. Otherwise, clinical isolates of HSV can be sent straight to a laboratory for testing without the need for further steps. They can be accessed as frozen isolates kept up at 70°C or as newly isolated viruses displaying at least 50% CPE in urbane cells, whichever is more appropriate.
The infected tissue culture tube should be thoroughly filled out with a suitable medium, such as modified Eagle's minimal essential medium (EMEM) supplemented with 2 milligrams of glutamine and 2 percent foetal bovine serum, when presenting fresh isolates (FBS). The tube should carry to the laboratory at room temperature, with the cap securely fastened and covered with parafilm to prevent contamination. In the Antiviral testing lab, all isolates should be regularly passed once and regrown to 50–100 percent CPE in culture tubes of the same cell type after they are inward. Primary rabbit kidneys, primary human neonatal kidneys (including those from newborns), mink lung, continuous African Green monkey kidneys (CV-1 or Vero), continuous human lung carcinoma (A-549), and human diploid fibroblasts (MRC-5 or WI-38) are among the cell types that can use for this purpose. CV-1 cells chosen because this cell line employs in the antiviral susceptibility experiment, which is why it is not compulsory. It is optional that all HSV isolates be at the first or second passage level before doing susceptibility; continual sub passage should be presented prior to testing. Harvesting and freezing aliquots of each isolate at a temperature of 70°C for future use. Antiviral Testing Lab & GeneticsThere are a number of indicators that may use to identify various viral strains. Temperature sensitivity, plaque size, antiviral sensitivity, and restriction endonuclease cleavage patterns among the characteristics that can be observed. It is not known what the molecular basis for most of these strain differences is, but viruses that are resistant to acyclovir usually have mutations in their thymidine kinase gene, while other resistant strains have mutations in their DNA polymerase gene. The molecular basis for most of these strain differences is unknown. The genome of the archetypal laboratory strain VZV Dumas is 124 884 base pairs (bp) in length. Many viral genes were discovered by comparison with HSV-1 genes that had similar sequences, as well as through genetic complementation experiments in which cell lines expressing specific VZV proteins were made used to enhance the development of HSV-1 mutants in the laboratory. It has been built to make targeted deletion, insertion, or site-direct point mutations in specific viral gene products using cosmids and bacterial artificial chromosomes (BACs) produced from the Zika virus (VZV). Also read: jis l 1902 Test Method Still Relevant in 2021? The discovery of microorganisms dates back to the 17th century by two members of the Royal Society, one being the Father of microorganisms Antoni van Leeuwenhoek. The most common ones among the huge lot are bacteria, viruses, fungi, Archaea, etc. One of the first life forms on Earth, Bacteria, which is a few micrometers long, is a type of biological cell consisting of prokaryotic microorganisms. Anti-bacterial testing refers to the process of observing the degree of resistance created by an anti-bacterium against bacteria. Prontosil was the first anti-bacteria to be discovered. Anti-bacterial testing reveals the level of effectiveness an antibacterial drug can achieve against a certain bacteria. The main concept of anti-bacteria is to make the environment too bad for the bad bacteria to survive.
About ISO 22196 or (JIS) Z 2801:- The iso 22196 jis z 2801 is the standard established which specifies the procedure of testing the surface of plastic, non-porous and intermediate product to inhibit the growth of bacteria. The (JIS) Z2801 is a test that inhibits the growth of microorganisms and has a lot of real-world applications ranging from clinics and hospitals to laboratories. It has become the standard test in the industry for antimicrobial tests on hard surfaces like plastic, ceramics etc. it tests how far the hard surfaces can inhibit the growth of the microorganisms in a 24 hour contact period. The (JIS) Z 2801 test has also achieved international accredits and is referred to as the International Organization for Standardization (ISO) procedure, iso 22196 jis z 2801. Procedure for conducting the test:- There are various available methods for effectively conducting the test like absorption, evaluation, bacterial suspension, Colony plate count, and Adenosine Triphosphate Luminescence methods. But the general testing procedure is as follows:- In the first step, the preparation of microorganisms in a liquid-cultured medium takes place with the growth of two bacteria Escherichia Coli and Staphylococcus Aureus. In the next step, the sterilization takes place with the concentration including 70% Ethanol. In the third step, Inoculation takes place, in which the microorganisms are introduced to the control and the testing surfaces. Then, after encouraging spreading, preventing evaporation, and getting accurate test results, the inoculum is covered with a sterile film. After determining the microbial concentration, it is ensured that this (JIS) Z 2801 method effectively neutralizes the anti-bacteria agent at the surface to be tested. It is then placed in a humid environment and left undisturbed to incubate after completing the other procedures. Then at the end, the microbial concentrations are determined once again and compared with the previous determination to evaluate the reduction of the microorganism. Then, the plate counting is done. Plus points of the test:- In the ISO 22196 method, the most striking feature is if the inoculum is not spilt beyond the test surface, then the results can be produced again and again without making any changes in the constitution. After completing the task, it shows a pass or fails criteria that remove the chances of discretion. It works for both bacteriostatic (inhibiting growth) and bactericidal (bacteria-killing) properties. Conclusion:- The (JIS) Z 2801 is the procedure that comes with drawbacks like it might not show the actual bacterial concentration. Still, there are various solutions via which accurate results can be obtained. Thus, the ISO 22196 is an excellent and effective method used globally, from clinics to labs. Also, Read - A closer view to the microbial reaction of Plastics What Is Antiviral Activity?
Finding an exact definition of scientific terms like Antiviral Activity can be challenging, and even if you find one, it will probably be hard to understand. Instead, let's look at a simple explanation of the term. Antiviral Activity is the extent to which a particular sample kills viruses or inhibits their ability to reproduce by suppressing the ability to replicate, which makes most viruses so dangerous. You can find good examples of Antiviral Activity almost anywhere: many natural plants, like rhubarb and sage leaf, offer intense antiviral benefits. On the other hand, textiles, fabrics, clothes, and different types of protective wear can also display Antiviral Activity by being made of materials that suppress the growth of viruses. How Do You Test For Antiviral Activity? The methods used for antibacterial test differs slightly based on the antiviral testing laboratory you contract. Still, in the end, they have to follow International Standards (ISO) to ensure that they get an accurate result. Another fact that introduces differences in the process is the sample being tested. A typical example would be textile samples, which are tested the following way:
Viruses and Organisms Used For Testing Antiviral Activity Antibacterial testing is always more dangerous than other bacteria activity tests because scientists have to be very careful with the virus samples. Still, to provide their clients with the best services, Antiviral Activity Testing labs today can test different kinds of samples for many organisms, like:
Conclusion If you're looking for an antiviral testing laboratory today, the first thing you should check is what tests they can perform for you. Labs that provide high service quality will perform ISO Certified tests like ISO18184 and EN 14476, using cell lines like MRC-5 cells, 229E, and MDCK cells for different virus tests. While checking up on technical requirements, ensure that you get a lab that combines good service quality and reasonable pricing! Also read - Is your textile business AATCC 100 tested? JIS L 1902 is a very popular antimicrobial product test. Antimicrobial product testing and performance generally represent a design that normally gets featured in the finished products. It is mostly related to a product’s ability to control and resist the growth of microorganisms in a particular product. The textile and the fabrics industries are the most regular customers of this test. The test is completely made for the textile and the fabrics to control the microorganisms in it. This involves the whole concept and procedures, which will help you understand the antimicrobial's nature in every product.
Knowing more about the test- Talking about JIS L 1902, it is very much popular worldwide and frequently used for testing antimicrobial additives. It is specially designed to have full control over the growth and life span of the microorganisms. The antimicrobial additives first originated in the Japanese Industrial Standard, which is why its name, “JIS.” It first entered the Japanese market, and afterward, it faced the whole world. The method is very much used in the textile industry. As being approved by all the institutions, it is very much preferable by every test lab. As being originated from the country japan itself, you cannot find any faults because Japanese are very much précised in their work. It is also used in the fabric industries for bedding, furniture, and apparel. Types of JIS L 1902 test- If you check officially, there are 2 types of the test included in it. One is a qualitative test, and the other one is a quantitative test. Both of the tests are designed in their particular way to give you the results of both perspectives.
Taking about the regular customers, the customers mostly choose the quantitative method over the qualitative method of the test because of the most common two bacteria, Staph and Kleb. Apart from this, the other organisms can be used as per the choice of the customers. The test microorganisms are completely tailor-made by getting growth in a fully liquid culture medium. The control and the test fabrics are generally inoculated with the microorganisms in triplets. It helps ensure that the inoculum is the only factor left with the fabrics which are left. Conclusion- JIS L 1902 is a test that is mostly used by the textile and fabrics industries. The whole test controls the growth and lifespan of the microorganisms in it. It is very beneficial for the products, and the customers do this test regularly in two different ways. Also read - Different Tests conducted at the antiviral testing laboratories. Testing is required to prove the Antibacterial qualities of a final product textile and support marketing claims or studies. Of course, laboratory testing is not the same as real-world testing. Therefore, they should compare results to real-world outcomes.
In the Antibacterial testing lab, Textiles are tested using a variety of conventional procedures, including but not limited to:
This test is carried out on both treated and untreated test material samples. The test sample is infected with 1 ml of test organism suspension containing roughly 1000,000 CFU/ml, per the AATCC 100 test procedure. Organisms are removed at a specific time using a diluted and plated neutralizer medium.This procedure evaluates the Antibacterial test agent's bacteriostatic and bactericidal properties.The hydrophobic nature of textiles might influence the Antibacterial activity test findings obtained by the AATCC 100 test technique.
This test method is used to determine if specimens treated with a non-leaching Antibacterial agent are resistant to microbe growth under dynamic contact circumstances. It is used to analyze substrate-bound Antibacterials and was developed for regular quality control and screening testing. All samples are shaken in dynamic shake flasks in a wrist action shaker to maintain good contact between the bacteria and the treated fiber, fabric, or other substrates throughout the test time by continual agitation of the test specimen in a bacterial suspension. Antibacterial agents that have been immobilized, such as surface-bound compounds, cannot disperse into their surroundings under normal operating circumstances. During the test time, the test material is constantly agitated in a bacterial solution, ensuring good contact between bacteria and the treated fiber (1hour). Microbial concentrations in treated and untreated samples are generally measured after 0, 1, or 24 hours of contact.
It is dependent on the intended purpose and the environment in which will utilize the textile product. Customers have the option of using one of the following methods to determine antibacterial activity: The bacterial suspension is injected directly onto the samples in the absorption technique. The Adenosine Triphosphate Luminescence technique and the Colony Plate Count method are both described for measuring bacterial enumeration. Also read - What does ISO 20743 test method mean for a common textile businessman ISO 22196 is an antimicrobial surface test that measures antibacterial activity on plastic and other non-porous surfaces. Based on JIS Z 2801, ISO 22196 is a surface test commonly used with standard bacteria that can be replaced depending on customer requirements. The ISO 22196 standard test method is designed to test plastic capabilities and other similar materials to kill or prevent the growth of microorganisms during the 24-hour contact period. Standard antimicrobial testing for ISO 22196 opposed both organisms, S. aureus and E. coli.
ISO 22196 is one of the most commonly asked antimicrobial surface test methods to evaluate the antimicrobial activity of antibacterial plastic products, including between products. ISO 22196 for concrete is adapted to test antimicrobial performance from concrete treated and not treated. Antimicrobial plastic can meet ISO 22196 requirements. However, when conditioned in a simulation environment, the treated plastic may not continue to work as referred to. It is important to incorporate environmental conditions in testing, such as UV exposure, moisture, and release agents that can affect the efficacy of antimicrobial plastic. ISO 22196 test strength
Weakness of the ISO 22196 test The ISO 22196 method does not necessarily represent the actual surface contamination event. The relatively dilute liquid microbial inoculum is spread over a large enough surface area and then wet guarded (usually for 24 hours). Most of the time, microbial contaminants dries quickly to the surface. It limits the time that juicy media is available to facilitate the interaction between antimicrobial surfaces and microorganisms. This means that ISO 22196 is a "best case" test type for many products. Also read - What is ISO 18184 testing? Here is all you need to know about it. Published by the American Society for Testing and Materials (ASTM), astm e2149 describes antibacterial activity properties under dynamic contact conditions and how antibacterial agents are tested. The official names of this standard are astm e2149. Standard test method for measuring the antibacterial activity of antibacterial agents under dynamic contact conditions.
Immobilized antibacterial agents are chemically bound and cannot freely diffuse into the environment under normal use conditions. Therefore, fiber test methods such as AATCC147-2004, which directly rely on the easy exudation of antimicrobial agents from treated fabrics, are unsuitable for assessing immobilized antimicrobial agents. This test method ensures good contact between the bacteria and the treated fiber, cloth, or another substrate by constantly stirring the test sample in the challenge suspension during the test period. The metabolic status of the challenge species can directly affect the measurement of the effectiveness of a particular antibiotic or drug concentration. In addition, the susceptibility of a species to a particular biocide can vary depending on its life stage (cycle). One hour of contact time in buffer allows for metabolic stagnation in the population. This test method standardizes both the growth conditions of the challenge species and the contact time with the substrate to reduce variability associated with the growth phase of the microorganism. Liquid analysis of the antibacterial activity of non-leaching agents provides the ability to wet the test substrate thoroughly. Wet surfactants can reduce the false negatives observed when comparing hydrophobic and hydrophilic substrates. ASTM E2149 test method This test is suitable for evaluating stressed or modified samples with proper control. This astm e2149 test method is designed to assess the resistance of antibacterial treated samples without leaching into microbial growth under dynamic contact conditions. This dynamic shaking flask test was developed for routine quality control and screening tests to overcome the difficulty of using classic antibacterial test methods to evaluate substrate-bound antibacterial agents. It was done. These difficulties include ensuring contact of the inoculum with the treated surface (for AATCC 100), the flexibility of recovery at various contact times, and improperly applied static conditions (AATCC 147. • The test also allows for various tests for hard water, proteins, blood, serum, multiple chemicals, other contaminants, or contamination due to physical/chemical stress or manipulation of the target sample. • Surface antibacterial activity is determined by comparing the results of test samples with controls performed concurrently. • The presence of exudative antibiotics is pre-determined and then determined by the presence of suppression zones. • This test method should only be performed by those who have been trained in microbiological techniques. • This standard may include dangerous goods, operations, and equipment. This standard is not intended to address all safety concerns associated with its use. Users of this standard are responsible for establishing good health and safety practices and determining the applicability of regulatory restrictions before use. |









 RSS Feed
RSS Feed
